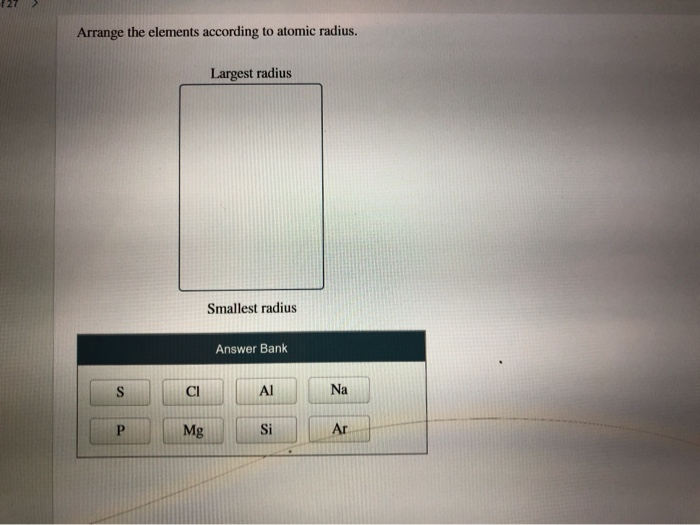 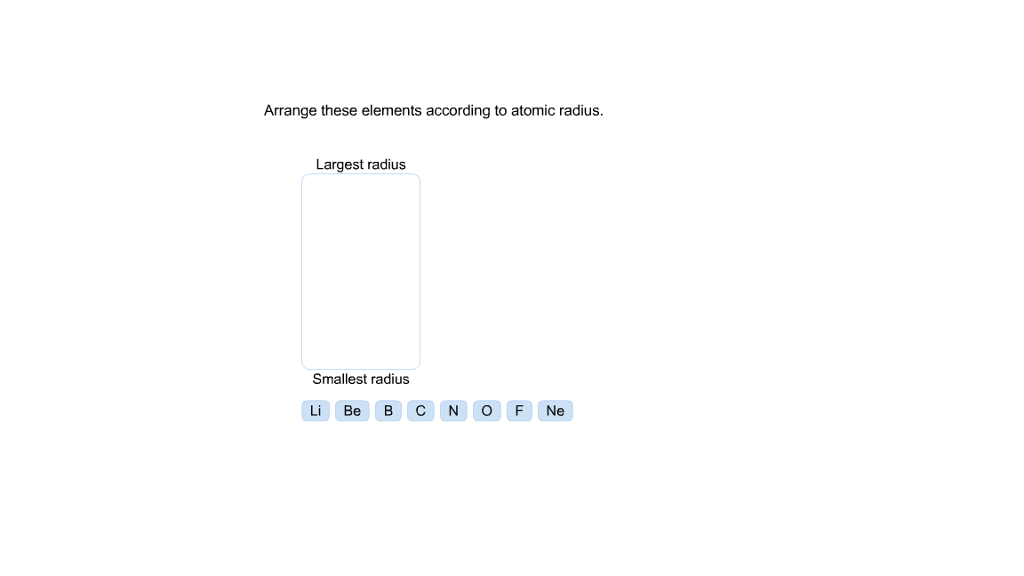
Calcium comes the last and forms the smallest ion. Its ion would be the largest among the five. A: Atomic radius of an element represents the distance of the nucleus from the outermost shell. Phosphorus comes the first among the five species and contains the least number of protons. Q: Arrange these elements in order of decreasing atomic radius: Cs, Sb, S, Pb, Se. The periodic table is arranged in the order of increasing proton number. 
A nucleus of strong positive charge pulls its electron cloud towards itself tightly such that the latter would have a small radius. Each proton carries a #+1# charge the more protons an atom or ion contains the stronger the attraction it poses on its electron cloud. We have to understand how Adams work and what it means when they have something appear in theory. Theyre going to be very close, given calling from India. So we need the periodic trend for atomic radio. We see that we have sodium, we have potassium, we have rubidium, we have cesium and we have lithium. And in our list of elements that we want to arrange. Positively-charged protons in the nuclei attract the negatively-charged electron cloud. All right, So this question fantastic to arrange this is Elektronik Siris in order of increasing atomic radius thes. And then we want to arrange these in order down to the smallest radius at the bottom. Similarly, a phosphorus atom ( #Z=15#) gains #color(navy)("three")# electrons to form a #"P"^(color(navy)(3-))# anion that contains #15+3=18# electrons. For example, a neutral calcium atom ( #Z=20#) has #20# electrons it loses #color(purple)("two")# of them to form a #"Ca"^(color(purple)(2+)# cation, which would thus contain #20-2=18# electrons. (a) F < N < Be < Li These elements are in same period and atomic radii decreases from left to right. Arrange the elements below in order of increasing radius. All five ions have an equal number of electrons and therefore share the same electron configuration. Elements in the periodic table are arranged according to their atomic.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
